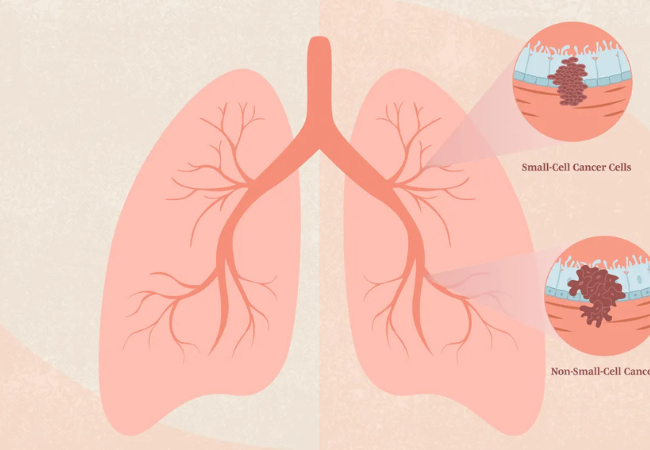
Metastatic non-squamous non-small cell lung cancer (NSCLC) is one of the most challenging forms of advanced lung cancer, requiring precision-based treatment strategies and timely access to innovative therapies. With the rise of targeted oncology medicines, patients today have access to advanced therapies designed for specific genetic mutations associated with NSCLC.
One of the emerging targeted therapies gaining attention globally is Hernexeos (Zongertinib), a next-generation treatment option for advanced or metastatic NSCLC.
For patients, caregivers, hospitals, and oncologists seeking access to advanced lung cancer medicines, Indian Pharma Network (IPN) helps facilitate the import of Hernexeos (Zongertinib) in India under the Named Patient Program (NPP).
What is Metastatic Non-Squamous NSCLC?
Non-small cell lung cancer (NSCLC) accounts for nearly 85% of all lung cancer diagnoses. Non-squamous NSCLC commonly includes adenocarcinoma and large-cell carcinoma subtypes.
Metastatic NSCLC refers to cancer that has spread beyond the lungs to other parts of the body, such as:
- Brain
- Bones
- Liver
- Adrenal glands
- Lymph nodes
Patients with metastatic non-squamous NSCLC often require advanced systemic therapies such as:
- Targeted therapy
- Immunotherapy
- Chemotherapy
- Personalized oncology treatment
Modern precision medicine has transformed NSCLC treatment in India by enabling therapies tailored to specific molecular mutations.
What is Hernexeos (Zongertinib)?
Hernexeos (zongertinib) is an emerging targeted therapy developed for advanced or metastatic non-squamous NSCLC associated with specific molecular and HER2-related alterations.
Zongertinib belongs to the category of next-generation tyrosine kinase inhibitors (TKIs) designed to target cancer-driving mutations more precisely.
As the latest NSCLC targeted therapy in India gains attention, oncologists are increasingly exploring advanced precision oncology approaches for difficult-to-treat lung cancers.
How Does Zongertinib Work in NSCLC?
Cancer cells often grow due to mutations in pathways responsible for cell growth and survival.
Hernexeos (Zongertinib) works by targeting specific molecular pathways involved in tumor progression and HER2-related signaling abnormalities.
By blocking these cancer-driving signals, Zongertinib may help:
- Slow tumor progression
- Reduce cancer cell growth
- Improve disease control
- Support personalized NSCLC treatment approaches
Targeted therapies like Zongertinib are becoming increasingly important in metastatic non-squamous NSCLC treatment in India.
Importance of Molecular Testing in NSCLC
Comprehensive biomarker testing is now considered essential in advanced lung cancer treatment.
Patients with metastatic NSCLC are often tested for:
- HER2 mutations
- EGFR mutations
- ALK rearrangements
- ROS1 mutations
- KRAS mutations
- MET amplification
- PD-L1 expression
Molecular testing helps oncologists determine whether targeted therapies such as Hernexeos (Zongertinib) may be suitable treatment options.
Precision diagnostics are playing a major role in shaping the future of NSCLC treatment in India.
Symptoms of Metastatic Non-Squamous NSCLC
Common symptoms may include:
- Persistent cough
- Chest pain
- Breathlessness
- Fatigue
- Weight loss
- Bone pain
- Coughing blood
- Neurological symptoms if cancer spreads to the brain
Early diagnosis and timely access to advanced targeted therapies remain critical.
Who May Benefit from Hernexeos?
Hernexeos (Zongertinib) may be considered for patients diagnosed with advanced or metastatic non-squamous NSCLC associated with specific molecular or HER2-related alterations, based on oncologist evaluation and molecular testing.
Patients seeking:
- HER2-mutated NSCLC treatment in India
- Advanced NSCLC targeted therapy
- Latest lung cancer medicines
- Precision oncology options in India
may discuss Zongertinib treatment possibilities with their healthcare provider.
Advanced NSCLC Treatment in India
India has become a growing center for advanced oncology treatment and global access pathways. Current NSCLC treatment approaches may include:
- Targeted Therapy: Precision medicines targeting specific mutations.
- Immunotherapy: Immune checkpoint inhibitors help the immune system attack cancer cells.
- Chemotherapy: Still widely used in combination regimens.
- Personalized Oncology Care: Treatment strategies customized according to molecular profiling.
As global oncology innovations continue expanding, access programs now play an increasingly important role for patients in India.
How to Access Hernexeos in India:
In some situations, specialty oncology medicines may not yet be commercially available in India. In this case, patients and hospitals may access such treatment through regulated pathways such as the Named Patient Program (NPP).
Named Patient Program (NPP) for Hernexeos in India:
The Named Patient Program (NPP) allows physicians to request access to internationally available medicines for patients with serious or life-threatening conditions when local treatment options may be limited.
Under this pathway:
- Medicines are imported for personal patient use
- Physician prescription and documentation are required
- Regulatory compliance procedures are followed
- Access is facilitated under applicable import regulations
The NPP pathway helps bridge the gap between innovation and patient access in oncology care.
Indian Pharma Network Support for Hernexeos Import
Indian Pharma Network (IPN) helps facilitate access to Hernexeos in India under the Named Patient Program.
IPN supports:
- Patients
- Caregivers
- Hospitals
- Oncologists
- Procurement teams
with services including:
- Global sourcing support
- Named Patient Program facilitation
- Regulatory documentation coordination
- International procurement assistance
- Cold-chain logistics support
- Timely medicine access coordination
As an oncology medicine supplier support platform in India, IPN helps streamline the process of accessing innovative cancer medicines globally.
Cost and Availability of Hernexeos in India:
This therapeutic drug is not yet available in India. The price of Hernexeos may vary depending on:
- Global supply conditions
- Import timelines
- Regulatory procedures
- Prescribed treatment quantity
- Country of sourcing
Patients and hospitals may contact Indian Pharma Network for the latest information regarding availability and import assistance.
Need Access to Hernexeos (Zongertinib) in India?
Indian Pharma Network assists patients, hospitals, and oncologists with:
- Named Patient Program support
- Import coordination for Hernexeos
- Global oncology sourcing assistance
- Regulatory documentation guidance
- International logistics support
- Access to advanced NSCLC medicines
For availability, import assistance, or treatment access support: Call: +91 96548 60915 or WhatsApp: +91 98104 69557.
Why Targeted Therapy is Transforming NSCLC Treatment
Targeted oncology medicines are changing the future of lung cancer management worldwide.
Benefits of precision oncology may include:
- Personalized treatment approaches
- Better mutation-specific targeting
- Improved disease management strategies
- Reduced impact on healthy cells
- Advanced treatment options for metastatic disease
As research evolves, therapies like Hernexeos (zongertinib) continue expanding the possibilities of metastatic NSCLC treatment in India.
Medical Disclaimer
This content is intended for informational and educational purposes only and should not be considered medical advice. Hernexeos (zongertinib) is a prescription-only medicine and should only be used under the supervision of a qualified healthcare professional. Treatment decisions must be based on physician evaluation, molecular testing, and applicable regulatory requirements.
References & Sources:
- American Cancer Society – Non-Small Cell Lung Cancer (NSCLC) Overview
- National Cancer Institute (NCI) – Non-Small Cell Lung Cancer Treatment
- World Health Organization (WHO) – Cancer Fact Sheet
- American Society of Clinical Oncology (ASCO) – Lung Cancer Guide
- National Comprehensive Cancer Network (NCCN) – NSCLC Guidelines
- U.S. Food and Drug Administration (FDA) – Targeted Cancer Therapies
- European Society for Medical Oncology (ESMO) – Metastatic NSCLC Guidelines
- PubMed – HER2-Targeted Therapies in NSCLC Research
- ClinicalTrials.gov – Zongertinib / HER2 NSCLC Studies
Is Hernexeos available in India?
Hernexeos (zongertinib) in India is not yet available but may be accessed through regulated pathways such as the Named Patient Program (NPP), subject to physician approval and regulatory compliance.
What is Zongertinib used for?
Zongertinib is an emerging targeted therapy being explored for advanced or metastatic non-squamous NSCLC associated with specific molecular alterations.
Can Hernexeos be imported in India?
Yes, Hernexeos may be imported in India under the Named Patient Program with required documentation and physician support.
What is the Named Patient Program?
The Named Patient Program allows access to internationally available medicines for patients with serious conditions when local access may be limited.
How long does Hernexeos import take?
Import timelines may vary depending on regulatory approvals, sourcing location, and logistics coordination.
Who can apply for Zongertinib access in India?
Access requests are generally initiated by treating physicians or hospitals for eligible patients.
Is Hernexeos approved for NSCLC treatment?
Treatment use and eligibility depend on country-specific approvals, physician evaluation, and molecular diagnosis.
How can Indian Pharma Network help?
Indian Pharma Network helps facilitate global sourcing, regulatory coordination, and Named Patient Program support for Hernexeos access in India