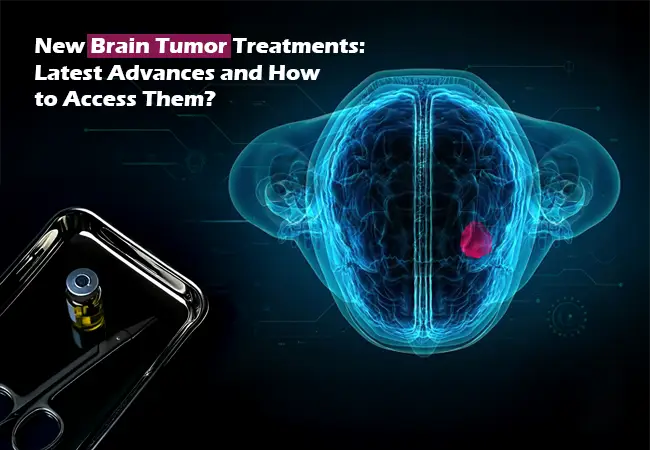
In both adults and children, brain tumors are considered one of the most complicated and life-altering conditions in oncology. The tumors can be classified as primary (originating in the brain) or secondary (spread from other parts of the body). The treatment for them is effective but complicated owing to the delicate region of the tumor, possible neurological complications, and multiple types of tumors present.
The most common types of brain tumors include:
- Glioblastoma
- Multiforme (GBM)
- Astrocytoma
- Oligodendroglioma
- Medulloblastoma
- Ependymoma
- Meningioma
- Brain Metastases (from other cancers)
The past few years have seen the emergence of new therapies, including targeted therapies, immunotherapies, tumor-treating fields (TTFields), and gene-directed therapies, which are significantly changing the treatment experience for patients diagnosed with high-grade or recurrent brain tumors.
Standard Treatments for Brain Tumors:
The first step in addressing an issue usually involves conducting an MRI or CT scan, procuring a biopsy, and conducting molecular analysis to ascertain the type and grade of the tumor. Treatment possibilities include:
- Surgery – Either partial or complete removal of the tumor.
- Radiation Therapy – Cancerous cells that remain after surgery are to be destroyed using radiation.
- Chemotherapy – Treatment via drugs either locally or throughout the body to stop the tumor from growing, can be done either systematically or locally.
- Targeted Therapy – Uses medication that prevents more specific changes in genes or proteins in the cells in the tumor.
- Immunotherapy – Treatment that helps the immune system of the patient stimulate and fight against cancer.
- Tumor Treating Fields (TTF) – Uses low-intensity electric fields to interfere with the reproduction of cancer cells.
Newly Approved Medicines and Treatments for Brain Tumors:
The following are the most cutting-edge treatments recently granted, which enhance the hope granted to people suffering from brain tumors, particularly those with aggressive ones or those most difficult to treat.
Temodar (temozolomide):
- Type: Alkylating Chemotherapy
Indication: Glioblastoma Multiforme (GBM), Anaplastic Astrocytoma - Mechanism: Crosses the blood–brain barrier to damage DNA in rapidly dividing tumor cells
- Approval: FDA-approved as first-line therapy for newly diagnosed GBM
Lomustine (CCNU):
- Type: Oral Nitrosourea Chemotherapy
- Indication: Recurrent high-grade gliomas
- Mechanism: Disrupts DNA replication and transcription in cancer cells
- Approval: Widely approved for use in relapsed gliomas; used off-label in combinations
Avastin (bevacizumab):
- Type: Monoclonal Antibody (Anti-VEGF)
Indication: Recurrent Glioblastoma - Mechanism: Inhibits vascular endothelial growth factor (VEGF) to cut off the tumor’s blood supply
- Approval: FDA-approved for recurrent GBM
Rozlytrek (entrectinib):
- Type: Tyrosine Kinase Inhibitor (TKI)
- Indication: NTRK gene fusion-positive solid tumors, including brain tumors
- Mechanism: Inhibits TRK fusion proteins involved in cancer growth
- Approval: FDA-approved for NTRK fusion-positive cancers with CNS involvement
Vitrakvi (larotrectinib):
- Type: Selective TRK Inhibitor
- Indication: NTRK fusion-positive CNS and solid tumors in adults and children
- Mechanism: Blocks TRK fusion proteins to halt tumor growth
- Approval: FDA-approved for NTRK fusion-positive tumors regardless of location
Dabrafenib + Trametinib:
- Type: BRAF + MEK Inhibitor Combination
- Indication: BRAF V600E-mutant gliomas (adult and pediatric)
- Mechanism: Blocks BRAF and MEK pathways to prevent tumor cell signaling
- Approval: FDA-approved for BRAF V600E-positive solid tumors, including CNS
Alymsys (bevacizumab)
- Type: Monoclonal
- Antibody (Anti-VEGF Therapy)
- Indication: Colorectal cancer, non-small cell lung cancer (NSCLC), glioblastoma, renal cell carcinoma, cervical cancer
- Mechanism: Inhibits vascular endothelial growth factor (VEGF), preventing tumor blood vessel formation and restricting tumor growth
- Approval: FDA-approved for multiple cancers, including recurrent glioblastoma and metastatic colorectal cancer
Mekinist (trametinib)
- Type: MEK Inhibitor (Targeted Therapy)
- Indication: Melanoma, non-small cell lung cancer (NSCLC), anaplastic thyroid cancer (with BRAF mutation)
- Mechanism: Inhibits MEK1 and MEK2 proteins in the MAPK pathway, blocking tumor cell growth and proliferation
- Approval: FDA-approved for BRAF V600 mutation-positive cancers, often used in combination with dabrafenib
Ojemda (tovorafenib)
- Type: RAF Kinase Inhibitor (Targeted Therapy)
- Indication: Pediatric low-grade glioma (LGG) with RAF alterations
- Mechanism: Targets RAF kinase pathway alterations, inhibiting tumor cell signaling and growth in RAF-driven tumors
- Approval: FDA-approved for pediatric patients (≥6 months) with relapsed or refractory low-grade glioma harboring RAF gene alterations
Voranigo (vorasidenib)
- Type: IDH1/IDH2 Inhibitor (Targeted Therapy)
- Indication: Low-grade glioma with IDH1 or IDH2 mutations
- Mechanism: Inhibits mutant IDH enzymes, reducing oncometabolite (2-HG) production and slowing tumor progression
- Approval: FDA-approved for adult patients with IDH-mutant low-grade glioma following surgery
Accessing New Brain Tumor Treatments in India and Other Countries:
Unfortunately, many of the above-listed medicines are not yet approved or widely available in various countries. Through Named Patient Programs (NPPs) and regulatory-compliant import pathways, patients and doctors can legally access these critical treatments.
Indian Pharma Network (IPN) is India’s Leading Partner in Named Patient Access and Import of Rare & Hard-to-Find Medicines.
We facilitate the supply of advanced brain tumor therapies like, Avastin, Rozlytrek, and others based on a valid prescription and regulatory permission.
If you or a loved one has been diagnosed with a brain tumor and urgently needs access to a newly approved or investigational treatment not available in your country, Indian Pharma Network (IPN) is ready to support you. Contact us to explore your brain tumor treatment options today.
Disclaimer: This content is for informational purposes only and does not replace medical advice. Always consult your treating physician before starting any new medicine or therapy.
We do not import or supply medical devices. We assist only with the import of prescribed medicines that are not available or approved in India but are approved in international markets, in accordance with applicable regulations.
How can I access new brain tumor treatments in India or other countries?
Indian Pharma Network (IPN) legally helps import medicines through Named Patient Programs which are not available in your country.
Can I buy Avastin for brain tumors in India?
Yes, with a valid prescription, we can assist in sourcing Avastin, and other approved or investigational brain tumor therapies from certified global suppliers.
What is the price of Temozolomide or Rozlytrek in India?
Prices of these therapeutic drug may vary depending on the manufacturer, dosage, and country of origin. Contact us today for the most accurate and updated quote.
Are these drugs available globally through IPN?
Yes. Indian Pharma Network (IPN) supplies to more than 150 countries through legal channels and supports patients across the world in accessing the newest cancer medicines.
Do you offer cold-chain shipment for brain tumor therapies?
Yes, we ensure cold-chain validated delivery for all temperature-sensitive medicinal products to maintain their potency and safety during transport.
Why Choose Indian Pharma Network (IPN)?
- Biggest Indian Company in the
- Named Patient Supply Segment
- 30+ Years of Global Pharma Access Experience
- 10,000+ Patients Served Across 150+ Countries
- 15,00,000+ Product Lines Facilitated
20+ Therapeutic Areas Supported, including oncology and neurology - Cold Chain Handling of Specialty Medications
- Access to US FDA, EMA, UK MHRA & WHO-GMP Certified Drugs
CDSCO & DGFT Regulatory Compliance for Import into India