Blincyto contains the active ingredient Blinatumomab. It is a bispecific T-cell engager (also called BiTE) immuno-oncology therapy, designed to target CD19 surface antigens on B-cells. The bispecific T-cell engager molecules fight cancer by assisting the body’s immune system to find out and target unhealthy cells by engaging T cells to cancerous cells. By bringing T cells close to cancerous cells, the T cells can inject toxins and trigger cancer cell death (apoptosis).
US Food and Drug Approval (FDA) approval: Blincyto injection was granted breakthrough therapeutic option and priority review designations by the US FDA and is approved for the treatment of:
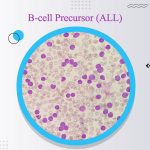
- CD19+ B-cell precursor ALL (B-ALL) in first or second complete remission with minimal residual disease/MRD greater than or equal to 0.1% in adults and pediatric patients.
- relapsed or refractory CD19+ B-cell precursor ALL (B-ALL) in adults and pediatric patients.
European Union (EU) approval: In the EU, the medication Blincyto is approved as monotherapy for the treatment of:
- adult individuals with Ph-negative (Ph-) CD19+ positive relapsed or refractory B-precursor acute lymphoblastic leukemia (ALL).
- adult individuals with Ph-negative (Ph-) CD19+ B-precursor ALL in first or second full remission with MRD/minimal residual disease greater than or equal to 0.1%.
- pediatric patients aged 12 months or more with Ph-negative (Ph-) CD19+ B-precursor ALL which is refractory or in relapse after being treated with at least 2 previous therapies or in relapse after previously taking allogeneic hematopoietic stem cell transplantation.
Blincyto Price in India:
Blincyto injection price in India is not fixed by the respective authorities because the medication is yet to be approved/registered/available in India. The medication Blincyto is commercially supplied as 38.5 mcg vials. In India, Patients, Clinicians, or Hospitals can approach trusted providers with extensive experience in importing Blincyto vials through legal channels. One can connect with Indian Pharma Network (IPN) via e-mail at info@indianpharmanetwork.in or call on +91 9310090915 to know more about our import procedure.
Reference:
https://www.blincyto.com/