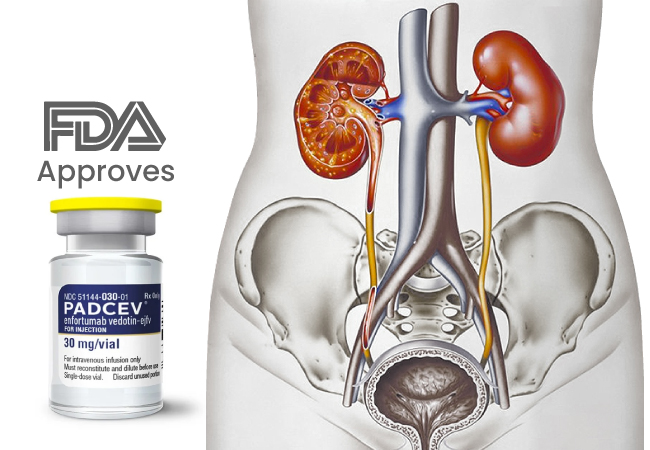
The FDA’s recent approval on 15 december 2023 of the enfortumab vedotin-ejfv (Padcev, Astellas Pharma) and pembrolizumab (Keytruda, Merck) combination marks a significant breakthrough for patients with locally advanced or metastatic urothelial cancer (la/mUC), particularly those ineligible for cisplatin-containing chemotherapy.
Based on data from the EV-302/KN-A39 (NCT04223856) trial involving 886 patients, this combination demonstrated superior outcomes compared to platinum-based chemotherapy. Notably, the median overall survival (OS) for the enfortumab vedotin-ejfv with pembrolizumab group was 31.5 months, a substantial improvement over the 16.1 months observed in the platinum-based chemotherapy group. Similarly, median progression-free survival (PFS) showed a noteworthy increase at 12.5 months versus 6.3 months, respectively.
Common adverse reactions, observed in over 20% of patients, included increased aspartate aminotransferase, rash, peripheral neuropathy, fatigue, diarrhea, and nausea. This emphasizes the need for careful monitoring and management of these side effects during treatment.
Healthcare professionals are advised to follow the recommended doses—1.25 mg/kg for enfortumab vedotin-ejfv and 200 mg for pembrolizumab—with adjustments for patient weight. The treatment involves intravenous infusions over specified intervals until disease progression or unacceptable toxicity.
This approval, granted under Project Orbis, a collaborative initiative between the FDA, Australian Therapeutic Goods Administration, and Health Canada, streamlines the submission and review process. The Real-Time Oncology Review pilot program and Assessment Aid facilitated a faster review, resulting in approval five months ahead of the FDA goal date.
Acknowledging the urgency and significance of this therapy, the FDA granted priority review and breakthrough designation, aligning with the agency’s expedited programs for serious conditions. Healthcare professionals are encouraged to report any adverse events associated with this treatment to the FDA’s MedWatch Reporting System.
This approval underscores the commitment to advancing oncology treatment options and signifies a crucial milestone in the fight against urothelial cancer. The Oncology Center of Excellence continues to lead the way in facilitating collaborative efforts for the benefit of patients worldwide.