Key Takeaways
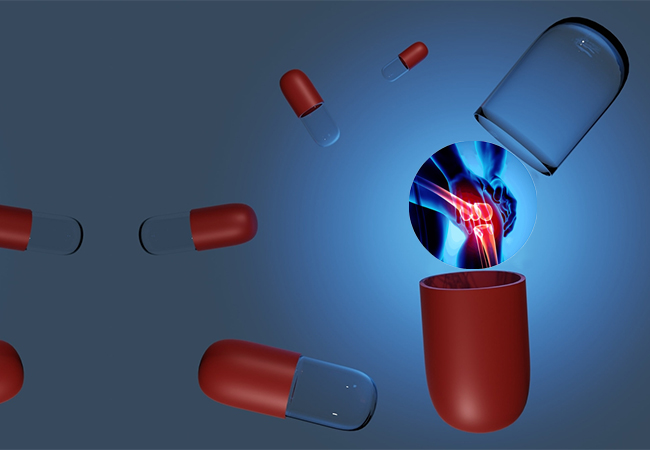
- Psoriatic arthritis (PsA) is a chronic, autoimmune form of arthritis that causes joint inflammation and occurs with psoriasis.
- The medicine company UCB recently developed a new Psoriatic arthritis (PsA) treatment known as Bimzelx.
- Bimzelx (bimekizumab) appears to be promising even in those who have not previously responded to treatment
- The therapeutic drug Bimzelx (bimekizumab) is now approved in multiple countries.
Overview of Psoriatic arthritis (PsA):
Psoriatic arthritis or PsA, is an immune-mediated musculoskeletal condition that affects roughly 30% of individuals with a history of psoriasis. The common symptoms of PsA are pain, swelling, and stiffness in joints as well as swollen fingers/toes, small pits in nails, nails changing color, and feeling extremely tired. PsA can impact people’s quality of life (QoL) and their day-to-day activities.
When someone develops the condition, healthcare providers initially prescribe conventional synthetic disease-modifying antirheumatic drugs (DMARDs). However, if these medicines do not work well, the clinicians may switch to (or add) biologic DMARDs, such as TNFi-IR. These drugs are highly specific and target a specific immune system pathway to scale down inflammation.
While these medicines often relieve symptoms and slow the disease’s progress, not everyone responds to the current medicines, and even individuals who do are often left with ongoing symptoms.
Role of Bimzelx in Psoriatic Arthritis (PsA):
Interleukin-17 (IL-17) is a group of pro-inflammatory cytokines that play a crucial role in both psoriasis and Psoriatic arthritis (PsA). The drug Bimzelx (bimekizumab) is a monoclonal antibody that selectively blocks two members of the IL-17 family: IL-17A and IL-17F.
These cytokines are developed from the same immune cells. Researchers believe they are particularly vital in driving the damaging inflammation linked with Psoriatic arthritis (PsA).
Research has demonstrated that both IL-17A and IL-17F are overexpressed in the skin and joints of those with psoriasis and Psoriatic arthritis. Bimzelx is the first approved treatment for Psoriatic arthritis (PsA) that selectively blocks IL-17A and IL-17F.
Study Insights:
The first study, named BE OPTIMAL, enrolled a total of 852 participants with Psoriatic arthritis (PsA) who had never been treated with biologic DMARDs.
The second study, called BE COMPLETE, enrolled a total of 400 participants with Psoriatic arthritis who had not responded well to one or two TNFi-IR biologics.
In both studies (BE OPTIMAL and BE COMPLETE), researchers said that those who were given Bimzelx had significantly better outcomes than those who were given a placebo.
Researchers said the medicine reduced the symptoms of Psoriatic arthritis (PsA) and stopped structural damage. Alongside clinical measures, individuals self-reported enhancements in physical function, pain, and fatigue.
They said the results of the study BE COMPLETE are promising for those for whom first and second-line medicines have failed. The medicine Bimzelx gives these individuals another treatment option. Also of note, the study demonstrated that the medicine`s beneficial effects became apparent after just 4 weeks.
Importantly, researchers said, the overall safety profile of Bimzelx was consistent with previous studies on existing medicines.
References:
https://www.bimzelx.com/
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(22)02302-9/fulltext
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(22)02303-0/fulltext
https://www.psoriasis.org/about-psoriatic-arthritis/#:~:text=Psoriatic%20arthritis%20(PsA)%20is%20a,related%20health%20conditions%20(comorbidities)